New Peer Reviewed Data Supports Use of Conformis Customized Knee Implants to Match Anatomic Variability in Patients
BILLERICA, Mass., April 23, 2019 (GLOBE NEWSWIRE) — Conformis Inc. (NASDAQ:CFMS), a medical technology company that uses its proprietary iFit Image-to-Implant technology platform to develop, manufacture and sell joint replacement implants customized to fit each patient’s unique anatomy, today announced the publication of peer reviewed data in the journal Clinical Orthopedics and Related Research which analyzed CT scan information from a population of over 24,000 patients. The study, “What Is the Possible Impact of High Variability of Distal Femoral Geometry on TKA? A CT Data Analysis of 24,042 Knees”, can be found at:
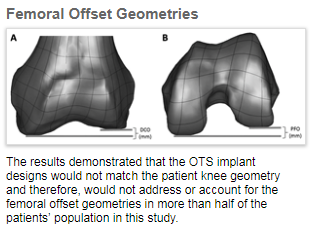
The results of this study quantified a high degree of anatomic variability within the study population in both distal (DCO) and posterior (PFO) femoral offsets (see pictures below), supporting the adoption of customized implant systems for a significant number of patients.
Publically available information on the design for standard or off-the-shelf (“OTS”) symmetric and fixed asymmetric implants was analyzed against the data derived from patient CT scans in this study. The results demonstrated that the OTS implant designs would not match the patient knee geometry and therefore, would not address or account for the femoral offset geometries in more than half of the patients’ population in this study.
There are two weight-bearing structures of the femur that articulate with the surface of the tibia: the medial and lateral femoral condyles. The native knee typically has differences in geometry of these femoral condyles both at the front, or distal elements (DCO), and on the back, or posterior elements (PFO), commonly referred to as condylar offset. Since OTS implants have a limited number of sizes, they cannot address these condylar offsets for patients knee implants.
In addition to the condylar offsets, the study investigators reviewed the dimensions of the knees for the patient population from the CT scans and found that, compared to the OTS available sizes, approximately one-third of the patients would have 3mm or more a size mismatch if the closest size was chosen. Size mismatch 3mm or more particularly when implants are too large has been linked to residual pain, which can be prevalent among women. Use of customized knee implants can minimize such size mismatching.
“Our results demonstrate the wide variability of knee anatomy among the general population,” said Professor Johannes Beckmann of SportKlinik Stuttgart, Stuttgart Germany. “One of the biggest challenges we face as knee surgeons is to make OTS implants fit into disparate patient anatomies. The data clearly shows that standard OTS implants, which come in fixed sizes and geometries, do not typically match that variability. In my experience the only implant which enables me to accommodate for all of the different anatomies I encounter in the operating room is the patient-specific Conformis knee.”
“This data supports what our engineers see on a daily basis when reviewing patient CT scans: a high percentage of anatomic variability exists within the general population,” said Mark Augusti, President and CEO of Conformis. “We believe this data contributes to understanding “why” a customized patient specific total knee implant provides improved patient comfort and kinematics as has been previously reported in our body of clinical evidence”.
Approximately 700,000 knee replacement procedures are performed annually in the US. This number is projected to increase to 3.48 million procedures per year by 2030 [1].
Approximately 160,000 knee replacement procedures are performed annually in Germany [2]
Disclosure: Johannes Beckmann, MD is a paid consultant to Conformis, Inc. and supports medical education of other surgeons.
About Conformis, Inc.
Conformis is a medical technology company that uses its proprietary iFit Image-to-Implant technology platform to develop, manufacture and sell joint replacement implants that are designed and manufactured to fit and conform to each patient’s unique anatomy. Conformis offers a broad line of patient conforming total and partial knee systems and a hip system that include sterilized single-use instruments delivered in a single package to the hospital. Conformis owns or exclusively in-licenses issued patents and pending patent applications that cover patient-specific implants and instrumentation for all major joints. In clinical studies, Conformis’ iTotal CR demonstrated superior clinical outcomes, including better function and greater patient satisfaction, compared to traditional, off-the-shelf implants.
For more information, visit www.conformis.com. To receive future releases in e-mail alerts, sign up at http://ir.conformis.com/.
Cautionary Statement Regarding Forward-Looking Statements
Statements in this press release about our future expectations, plans and prospects, as well as other statements containing the words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” or “would” and similar expressions, constitute forward-looking statements within the meaning of the safe harbor provisions of The Private Securities Litigation Reform Act of 1995. You should not place undue reliance on our forward-looking statements. Actual results could differ materially from the projections disclosed in the forward-looking statements we make as a result of a variety of risks and uncertainties, including risks and uncertainties described in the “Risk Factors” sections of our public filings with the Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent our views as of the date hereof. We anticipate that subsequent events and developments may cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date hereof.
[2] Analysis of Individual Inpatient Data From German and US Hospitals for the Years 2005 to 2011 Annelene Wengler, Dr. rer. pol.,1 Ulrike Nimptsch, Dr. rer. pol.,1 and Thomas Mansky, Prof. Dr. med.*,1
Related Posts
iTotal
The iTotal system offers two options that are both designed to restore the natural shape of a patient’s knee – iTotal CR and iTotal PS.
iTotalAbout Conformis
We start with a simple idea: make the implant fit the patient rather than force the patient to fit the implant
About Conformis