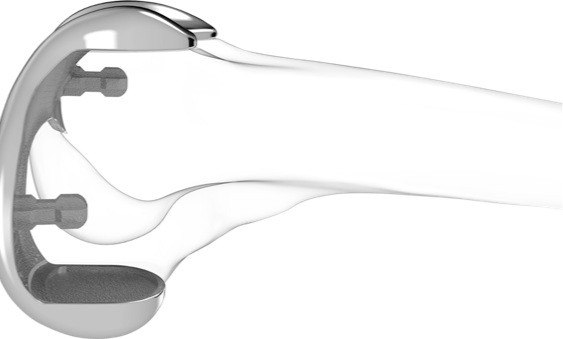
Outpatient Facilities/Ambulatory Surgery Centers & Conformis
In the wake of COVID-19, we know you will be under more pressure than ever before to treat a large number of patients as efficiently as possible. Demand for outpatient joint replacement procedures is growing and will remain high. Successful facilities benefit from a trusted and aligned team, method for patient selection and communication, and an ally dedicated to the overall success of their TKA practice.
Demand for outpatient joint replacement procedures is growing. Successful facilities benefit from a trusted and aligned team, method for patient selection and communication, and a partner dedicated to the overall success of their practice.
Interested in implementing Conformis at your ASC or outpatient facility? Click here.
This is where Conformis comes in.
Coverage
Conformis’ pre-navigated knee implant (Identity ImprintTM) and hip implants are covered under the same reimbursement codes as our competitors’ off-the-shelf implants. When patients choose to receive a personalized knee implant (iTotal® Identity), then those patients are responsible for paying an incremental, out-of-pocket personalization fee, which varies by medical facility.
![]()
A Comprehensive Approach to Address Cost Drivers
We understand your pain points
Outside the OR suite
- Instrument resterilization ~$60 per tray5
- Complicated inventory management
How Conformis can help
- 0-1 trays: reduced need for sterilization
- Decreased risk for resterilization errors
- Simplified ordering and delivery models
- Just-in-time delivery model: no implant inventory
We understand your pain points
Inside the OR suite
- Operating room costs ~$60 per minute1
How Conformis can help
- Very little setup
- Streamlined case prep
- Potential for decrease in skin-to-skin time
- Single-use patient‑specific instrumentation
- Decreased turnover time
We understand your pain points
After the procedure
- Discharge to rehab costs ~ $70002
- Average hospital complication ~ $31003
How Conformis can help
- Patients discharged faster4
- Less likely to be discharged to rehab5
- Fewer patients require blood transfusions5
- $913-$2,200 total cost of care savings4,5
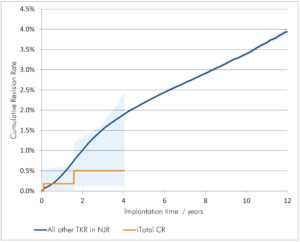
- Lower cumulative revision rate6
- No annual/monthly capital expenditure for robot or associated disposable instruments
Superior Clinical Outcomes
Studies comparing Conformis’ personalized knee implants to OTS TKAs have shown:
- Conformis patients discharged in under 24 hours, at a significantly higher rate than OTS patients1
- Fewer Conformis patients required blood transfusions2
- Fewer patients experienced adverse events at both discharge and 90 days after treatment2
- $913-$2,200 total cost of care savings1,2 to the payer
- Cumulative revision rate of 0.5% at four years compared to 1.9% cumulative revision rate at the same time-point among all knee replacement patients3
Outpatient Specific Training
Regional, local and virtual training opportunities are available:
- Surgeon Visitation Program: travel to established ASC facilities and scrub into cases, review protocols, tour the facility/
- National Training Labs: experience clinical discussions with peers during didactic sessions, hands-on bioskills session hosted by our experienced surgeon proctors
Increase Case Volume and Efficiency
Frequently Asked Questions


Unlike off-the-shelf (OTS) systems that require up to eight costly sterilized instrument trays, the Conformis model includes a single pre-sterilized kit of patient-specific instruments, a single reusable instrument tray, and no implant inventory.


Because Conformis implants are delivered as one box and one tray, there are fewer instruments to manage and the case itself requires very little set up. Studies have shown that medical devices having single package design with pre-sterilized instruments can help save 15-20 minutes of setup time7,8. The Conformis system uses single-use patient‑specific instrumentation, which is discarded after the procedure, decreasing turnover time.


Since beginning our Direct to Consumer program in 2017, we have drawn hundreds of thousands of patient prospects to our web site and patient call center, educating them, enabling them to find medical facilities and surgeons in their area who are trained in our technology, and connecting them with those surgeons.


Conformis is aware that today’s patients are digitally savvy and actively involved in their healthcare. Our highly targeted Direct to Patient programs span multiple channels (both traditional/print outlets and digital) to direct a high volume of qualified and educated patients to Conformis surgeons faster.
Click here to learn more about our Patient Engagement marketing programs
ASC and Outpatient Surgeon Testimonials
Gary Levengood, MD – Atlanta, GA
Wallace Huff, MD – Lexington, KY
1 Shippert, R., et al.; “A study of time dependent operating room fees and how to save$100,000 by using time –saving products”; Am Journ Cosm Surg; 2005; V22(N1):pp. 25-34
2 Ramos, N. L., Wang, E. L., Karia, R. J., Hutzler, L. H., Lajam, C. M., & Bosco, J. A. (2014). Correlation between physician specific discharge costs, LOS, and 30-day readmission rates: an analysis of 1,831 cases. The Journal of arthroplasty, 29(9), 1717-1722
3 Culler, S. D., Jevsevar, D. S., Shea, K. G., Wright, K. K., & Simon, A. W. (2015). The incremental hospital cost and length-of-stay associated with treating adverse events among Medicare beneficiaries undergoing TKA. The Journal of arthroplasty, 30(1), 19-25
4 Buch, R, et al.; “Does Implant Design Affect Hospital Metrics and Patients Outcomes? TKA Utilizing a ‘Fast-Track’ Protocol”; ICJR Pan Pacific; 2016
5 Culler, S. D., Martin, G. M., & Swearingen, A. (2017). Comparison of adverse events rates and hospital cost between customized individually made implants and standard off-the-shelf implants for total knee arthroplasty. Arthroplasty today, 3(4), 257-263.
6 Implant Summary Report for the iTotal G2 XE and iTotal G2 (Bicondylar tray). Beyond Compliance database February 11, 2018, pages 19-20
7 Takeuchi, H.; “Technical Report: Disposable Laparoscopic Surgical Instruments and the Economic Effects of Repackaging”; Journal of Lap & Adv. Surg Tech; Apr 2005
8 Watters, TS.; “Analysis of procedure-related costs and proposed benefits of using patient-specific approach in total knee arthroplasty”; J Surg Orthop Adv; 2011
iTotal
The only truly patient-specific cruciate retaining (CR) and posterior stabilizing (PS) total knee replacements.
iTotalAbout Conformis
We start with a simple idea: make the implant fit the patient rather than force the patient to fit the implant
About Conformis