Conformis Announces FDA Clearance and Full Commercial Launch of Next Generation Hip System at the 2019 AAHKS Conference
Designed to enhance operational efficiency for the surgeon, the Conformis Hip System is the most advanced, patient-conforming hip replacement system available
BILLERICA, Mass., Nov. 07, 2019 (GLOBE NEWSWIRE) — Conformis, Inc. (NASDAQ:CFMS), a medical technology company that uses its proprietary iFit Image-to-Implant technology platform to develop, manufacture, and sell patient-specific arthroplasty joint replacement implants designed to fit each patient’s unique anatomy, today announced FDA clearance of the Company’s next generation Conformis Hip System. The Company will be featuring the new 3D-designed Conformis Hip System as a key technology focus at the 2019 American Academy of Hip and Knee Surgeons annual conference November 7–10, 2019 in Dallas, TX.
Conformis previously announced the Company’s first hip surgeries started in July 2018 during its limited launch of the Conformis Hip System. The new system incorporates design improvements based on surgeon feedback after completion of over 400 surgeries.
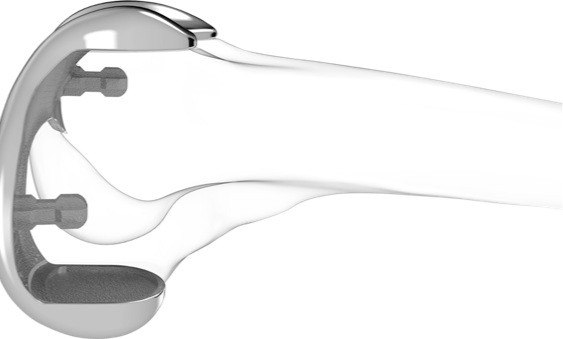
Similar to the design process for the Conformis knee technologies, the Conformis Hip System uses proprietary advanced imaging and design software, to deliver a patient-specific pre-operative surgical plan with a comprehensive set of iJig instruments and patient-matched implants.
“Our new Conformis Hip system is compatible with multiple surgical approaches and is designed to provide the surgeon an efficient and accurate experience in the execution of the operative plan,” said Mark Augusti, Chief Executive Officer of Conformis. “We believe our system will allow surgeons to improve efficiency in the OR, and importantly—for the anterior approach—potentially eliminate the use of fluoroscopy.”
The Conformis Hip System iJigs work in conjunction with anatomic landmarks to ensure the proper placement and orientation of the femoral stem, as well as the acetabular cup. In addition, Conformis’ proprietary reamer system provides a single-use, patient-matched reaming solution that delivers efficient and accurate cup orientation to a controlled depth for each surgical case.
The global hip joint reconstruction market is projected at over $7B and approximately 400,000 total hip replacements are performed in the United States each year.
About Conformis, Inc.
Conformis is a medical technology company that uses its proprietary iFit Image-to-Implant technology platform to develop, manufacture, and sell joint replacement implants that are designed and manufactured to fit and conform to each patient’s unique anatomy. Conformis offers a broad line of patient-conforming total and partial knee systems and a hip system that includes sterilized, single-use instruments delivered in a single package to the hospital. Conformis owns or exclusively in-licenses issued patents and pending patent applications that cover patient-specific implants and instrumentation for all major joints.
For more information, visit www.conformis.com. To receive future releases in e-mail alerts, sign up at http://ir.conformis.com.
Cautionary Statement Regarding Forward-Looking Statements
Statements in this press release about our future expectations, plans and prospects, as well as other statements containing the words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” or “would” and similar expressions, constitute forward-looking statements within the meaning of the safe harbor provisions of The Private Securities Litigation Reform Act of 1995. You should not place undue reliance on our forward-looking statements. Actual results could differ materially from the projections disclosed in the forward-looking statements we make as a result of a variety of risks and uncertainties, including risks and uncertainties described in the “Risk Factors” sections of our public filings with the Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent our views as of the date hereof. We anticipate that subsequent events and developments may cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we specificallydisclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date hereof.
Media Contact:
Investor Relations
ir@conformis.com
(781) 374-5598
Related Posts
CORDERA™ HipRx™
CORDERA™ HipRx™About Conformis
We start with a simple idea: make the implant fit the patient rather than force the patient to fit the implant
About Conformis