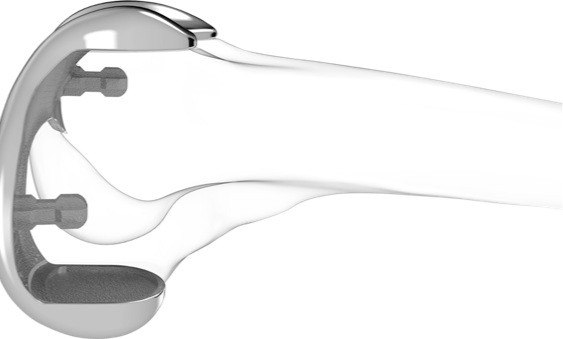
iUni & iDuo:Fully Personalized Partial Knee Replacements
iUni and iDuo are fully personalized or patient-specific partial knee replacement options for either unicompartmental or bicompartmental osteoarthritis of the knee. Both products offer customized femoral and tibial implant fit and shape, which gives the knee the potential to feel and move more naturally, and simple surgical technique.
iDUO Bicompartmental Knee Resurfacing System
The iDuo® G2 bicompartmental knee resurfacing system offers surgeons a unique option for early osteoarthritis intervention. The iDuo G2 offers a revolutionary alternative to traditional total knee replacement by combining proven TKA principles for patellofemoral treatment with the advantages of the Conformis partial knee replacement system. With the iDuo G2, a surgeon can treat just the affected compartments with a precise, patient specific implant that delivers unparalleled fit, preserves all ligaments and conserves far more bone than traditional TKA. Not only does this provide patients with more natural knee motion, it also preserves future treatment options—a benefit to both patients and surgeons alike.

Benefits of the iDUO Partial Knee Replacement System:

Personalized fit, natural kinematics:
- Unlimited femoral and tibial component shapes and sizes
- Anatomic shape and ligament preservation for natural kinematics
- Full coverage of all weight bearing areas
- Tibial component matched to cortical rim

Less traumatic, more bone preserving than TKR:

Simple, reproducible surgical technique:

Operating room efficiencies:
iUNI G2 Unicompartmental Knee Resurfacing System
Superior implant fit and performance requires a patient-specific approach to partial knee replacement. The Conformis Partial Knee Resurfacing Systems use proprietary technology and patient-specific imaging data to personalize an implant and instrumentation set for each patient.

Benefits of the iUNI Partial Knee Replacement System:

Patient-specific fit, wear-optimized design:
- Anatomic shape and fit for natural kinematics
- Full coverage of all weight bearing areas
- Tibial component matched to cortical rim
- Articulating surfaces engineered to reduce contact stress

Minimally invasive, minimally traumatic:

Simple, reproducible surgical technique

Operating room efficiencies
More Conformis Knee Replacement Products:
Healthcare Professionals
Resources and information for medical professionals
Healthcare Professionals


 iDuo Brochure
iDuo Brochure